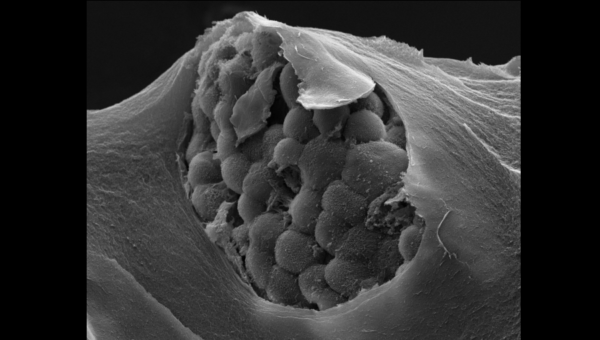
MRM Insights: A Case for Mechanically Defined Matrices

Dr. Joseph Matt Kinsella
Every month, in the MRM Insights, a member of the MRM Network writes about stem cells and regenerative medicine from a different perspective. This month, Dr. J. Matt Kinsella, Associate Professor in the Department of Bioengineering, presents a case for mechanically defined matrices.
A Case for Mechanically Defined Matrices
Scaffolds play a critical role in the physical support and contact-mediated guidance of cell migration, differentiation, and proliferation during tissue regeneration. Living cells are in a continuous exchange of momentum with their environment. Understanding how cells respond to external stimuli is a fundamental question in cell biology, with implications in development, collective cell migration or motility, or the emergence of disorders. The mechanical properties of scaffolds, including stiffness and porosity, are known to significantly impact how cells interact dynamically and reciprocally with their environment. Here we present, a simplified case to understand the relationship between a scaffold’s mechanical properties and biological outcome as a critical design criterion.
A detailed physical and mathematical approach to matrix design, with specific consideration of the ability of matrix materials to be used as cell-laden ‘bioinks’ for 3D bioprinting is presented in the following publications from our group [1,2]. In these examples, matrix elasticity is shown to play a critical role in cell viability, formation, and progression of spheroids [3, 4, 5].
The effect of elasticity on cell function relies on the expression of proteins, such as integrins, and the downstream focal adhesion complex proteins, which are sensed and transduced into mechanical and biochemical signals in specific pathways [4]. The necrotic core that develops internally within spheroids is dependent upon nutrient and oxygen transport limitations. Spheroids with diameters ranging from 200 to 500 μm are large enough to develop chemical gradients, while spheroids >500 μm develop a central secondary necrosis where the inner cells die by apoptosis or necrosis. Spheroids with diameters <150 μm are frequently used for drug testing and may be sufficient to exhibit 3D cell–cell and cell–matrix interactions but are not large enough to exhibit oxygen gradients with hypoxic regions or proliferation gradients. Balancing transport limitations with intrinsic matrix properties allows the control mechanisms to modulate the growth of cells, their proliferation rate, spheroid or organoid formation, and in the case of stem cells (as below) mechanisms to influence their differentiation.
What role does stiffness have on cell proliferation?
One of the most extensively studied relationships between scaffold mechanical properties and cellular behavior is the effect of scaffold stiffness on cell proliferation. The mechanical stiffness of the scaffold can be quantified by its modulus, defined as the ratio of stress to strain. Cellular proliferation is highly influenced by scaffold stiffness and differentiation pathways are defined in part by the stiffness of the niche or artificial scaffold.
Cells tend to exhibit greater proliferation rates on stiffer scaffolds relative to softer scaffolds. This relationship has been observed across various cell types, including mesenchymal stem cells (MSCs), human dermal fibroblasts, and osteoblasts [6]. This relationship can be modeled using a power law equation, where the cellular proliferation rate (P) is proportional to the scaffold stiffness (S) raised to a power law exponent (n) and multiplied by a constant (K). The equation can be expressed as:
P=KSn
Where n varies between cell types and scaffold materials.
What role does stiffness have on cell differentiation?
In addition to affecting cell proliferation, scaffold stiffness impacts cell differentiation. Cell differentiation refers to the process by which cells acquire specific functions and specialize in a particular tissue such as osteogenic differentiation of MSCs when the cells are cultured on stiffer scaffolds relative to softer scaffolds [7]. This relationship can be attributed to the regulation or activation of mechanical signaling pathways in response to scaffold stiffness [8]. The organization and interactions of the cytoskeleton and extracellular matrix, which are crucial components of these mechanical signaling pathways, are readily influenced by scaffold stiffness.
What role does stiffness have on extracellular matrix synthesis?
The mechanical properties of scaffolds also affect ECM synthesis, which is a critical factor in cell proliferation and differentiation. The ECM provides physical and biochemical cues to cells, including mechanical signaling pathways. Scaffold stiffness, and viscoelastic properties, have also been demonstrated to influence the synthesis of structural ECM proteins and ultimately their organization.
The deposition of collagen, a significant component of the ECM, is increased on stiffer scaffolds compared to softer scaffolds. This relationship can also be modeled using a similar form of the power law equation, where the amount of collagen deposited by cells (C) is proportional to the scaffold stiffness (S) raised to a power law exponent (n) and multiplied by a constant (K). The equation can be expressed as:
C=KSn
Where n varies between cell types and scaffold materials.
As 3D cell culture and bioprinted in vitro models gain acceptance interest in considering dynamic (mechano)reciprocity can not only be thought of by the addition of defined chemical factors but also by material selection.
References
[1] Jiang, Tao, et al. “Engineering bioprintable alginate/gelatin composite hydrogels with tunable mechanical and cell adhesive properties to modulate tumor spheroid growth kinetics.” Biofabrication 12.1 (2019): 015024.
[2] Jiang, Tao, et al. “Extrusion bioprinting of soft materials: An emerging technique for biological model fabrication.” Applied Physics Reviews 6.1 (2019): 011310.
[3] Guzman A, Ziperstein M J and Kaufman L J 2014 The effect of fibrillar matrix architecture on tumor cell invasion of physically challenging environments Biomaterials 35 6954–63
[4] Yeh Y C, Ling J Y, Chen W C, Lin H H and Tang M J 2017 Mechanotransduction of matrix stiffness in regulation of focal adhesion size and number: reciprocal regulation of caveolin-1 and beta1 integrin Sci. Rep. 7 15008
[5] Paszek M J et al. 2005 Tensional homeostasis and the malignant phenotype Cancer Cell 8241–54
[6] Steward, Andrew J., and Daniel J. Kelly. “Mechanical regulation of mesenchymal stem cell differentiation.” Journal of anatomy 227.6 (2015): 717-731.
[7] Titushkin, Igor, and Michael Cho. “Modulation of cellular mechanics during osteogenic differentiation of human mesenchymal stem cells.” Biophysical journal 93.10 (2007): 3693-3702.
[8] Arnsdorf, Emily J., et al. “Mechanically induced osteogenic differentiation–the role of RhoA, ROCKII and cytoskeletal dynamics.” Journal of cell science 122.4 (2009): 546-553.
Photo Credit: “Gastric patient tumor organoid within a mechanically-defined tissue-like matrix”, courtesy of Salvador Flores-Torres.
